
How to Calculate the Formal Charges for HCO3 (Bicarbonate ion ) YouTube
Lewis Structure of Carbonic Acid (H2CO3) The formula of carbonic acid is H2CO3. It has two H atoms, one C atom, and three O atoms. To understand the molecular formula of H2CO3, we have to observe the electronic configuration of the participating atoms and how many atoms they have in the outer shell.
[Solved] Write Lewis structures for these ions. (a) HCO3^ Bicarbonate ion
Frequency calculation at the HF/6-31G*//HF/6-31G* level showed that the structure 8 is not a minimum, as it contains two imaginary frequencies. $\ce{H3CO3+}$ shares structural similarities with its triaza-analog, the guanidinium ion, as both are possessing resonance stabilization via their onium forms [3, p. 60]. References
Hco3lewis Structure
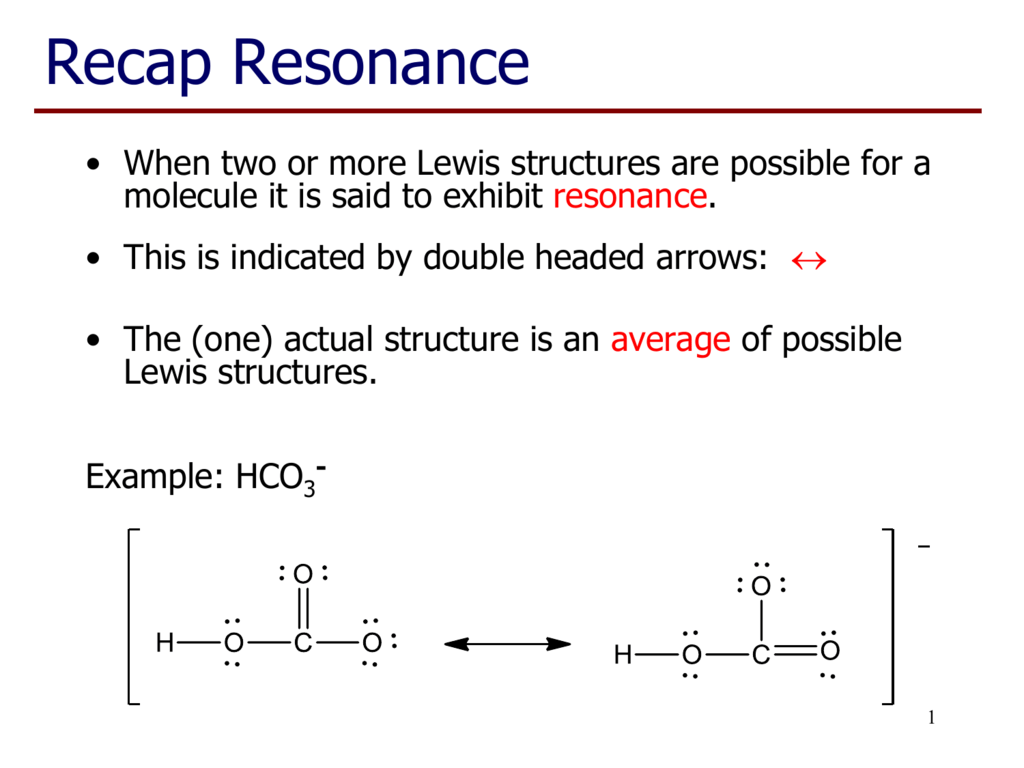
HCO3- Lewis structure November 7, 2023 by Deep The information on this page is fact-checked. HCO 3- Lewis structure HCO 3- (bicarbonate) has one hydrogen atom, one carbon atom, and three oxygen atoms. In the HCO 3- Lewis structure, there is one double bond and two single bonds around the carbon atom, with three oxygen atoms attached to it.
Lewis dot structure for hco3 examquiz
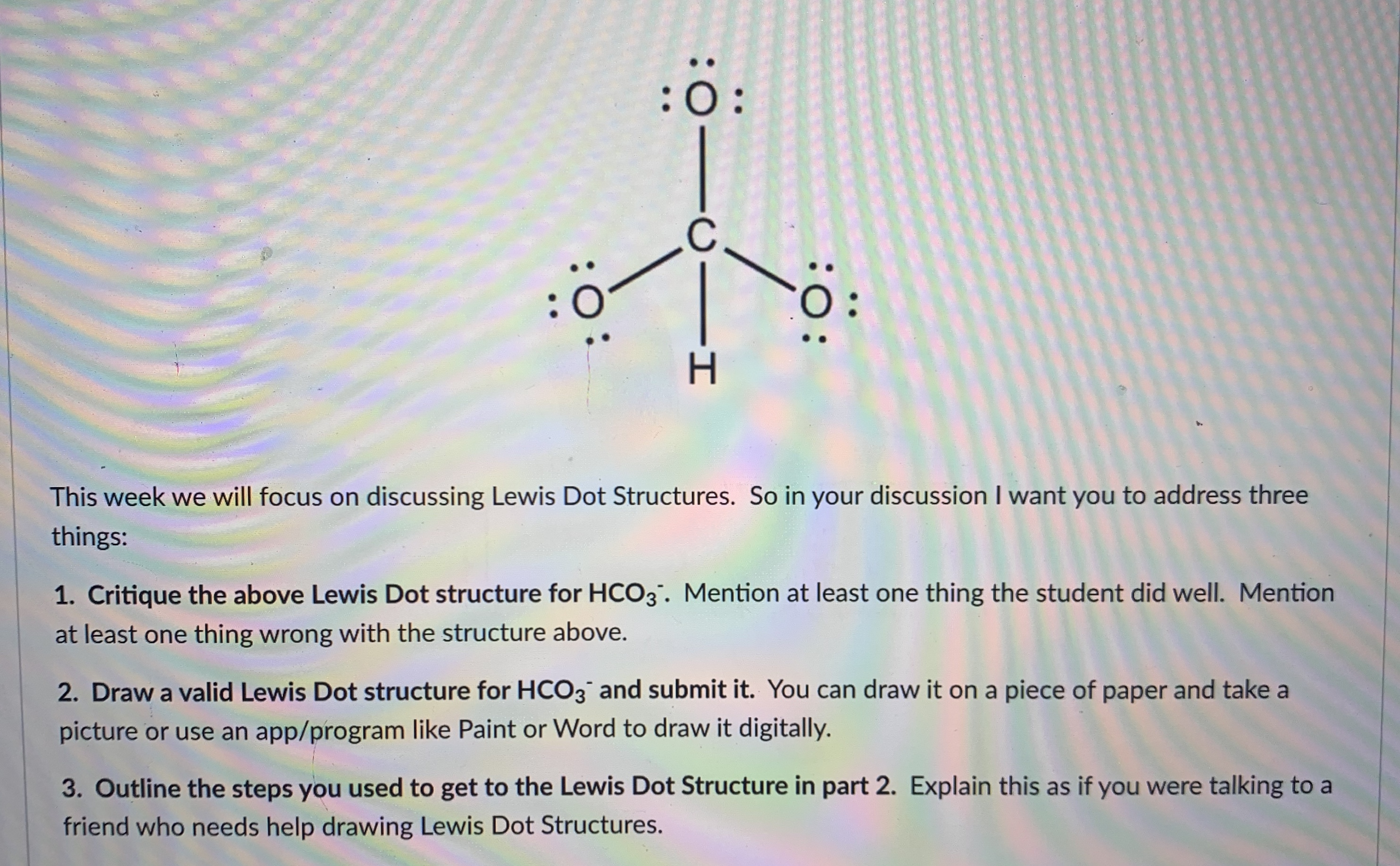
Lewis structure of HCO3- (Bicarbonate ion) contains one double bond between the Carbon atom (C) & one Oxygen atom (O) and the rest other atoms are single bonded with each other. The Carbon atom is at the center and it is surrounded by 2 Oxygen atoms and one O-H bond. The single bonded Oxygen atom has -1 formal charge.
Solved Which of the following is a correct Lewis structure
Subscribe 661 views 1 year ago Lewis Structure Hello Guys! In inorganic chemistry, bicarbonate is an intermediate form in the deprotonation of carbonic acid. It is a polyatomic anion with the.

HCO3 Lewis structure, Molecular geometry, Hybridization, Polar or
HCO3- lewis structure has a Carbon atom (C) at the center which is surrounded by two Oxygen atoms (O) and one O-H group. There is 1 double bond between the Carbon atom (C) & Oxygen atom (O) and the rest other atoms have a single bond. There is a -1 formal charge on the single bonded Oxygen atom (O).

Lewis dot structure for hco3 examquiz
By Sarnali Mukherjee HCO3- Lewis structure is reliable in denoting considerable chemical and physical properties of Bicarbonate. As Lewis structure brings forth a fundamental sketch of HCO3-, it is effective in highlighting the electronic fact about the compound.
SOLVED(a) The structure of the bicarbonate (hydrogen carbonate) ion
Bicarbonate (HCO3-) Ion Lewis Structure Iodate ion (HCO 3-) Ion Lewis Structure Bicarbonate ion contains one carbon atom, three oxygen atoms and one hydrogen atom. Lewis structure of carbonate ion (HCO 3-) contains one C=O bond, two C-O bonds and one O-H bond. There is -1 charge on one oxygen atom in HCO 3- lewis structure. HCO 3- lewis structure

Lewis Dot Structure For Sodium
HCO3- Molecular Geometry / Shape and Bond Angles Wayne Breslyn 725K subscribers Join Subscribe Subscribed 29K views 10 years ago A quick explanation of the molecular geometry of HCO3- including.
Draw a Lewis structure for the bicarbonate ion, HCO3^().
Drawing the Lewis Structure for H 2 CO 3. When we have an H (or H 2) in front of a polyatomic molecule (like CO 3, SO 4, NO 2, etc.) we know that it's an acid. This means that the Hydrogen atoms will be attached to the outside of the oxygen molecules. For the H 2 CO 3 Lewis structure (Carbonic Acid) make sure you put the Hydrogen atoms on the.
[Solved] Write Lewis structures for these ions. (a) HCO3^ Bicarbonate ion
In the lewis structure of carbonic acid (H 2 CO 3 ), carbon atom is the center atom and there are two -OH groups. Also, there is one double bond between carbon and oxygen atoms. As some molecules. there are no lone pairs on carbon atom. From H 2 CO 3 lewis structure, we can say H 2 CO 3 is a dibasic acid. In this tutorial, we will cover how to.
Lewis dot structure for hco3 examquiz
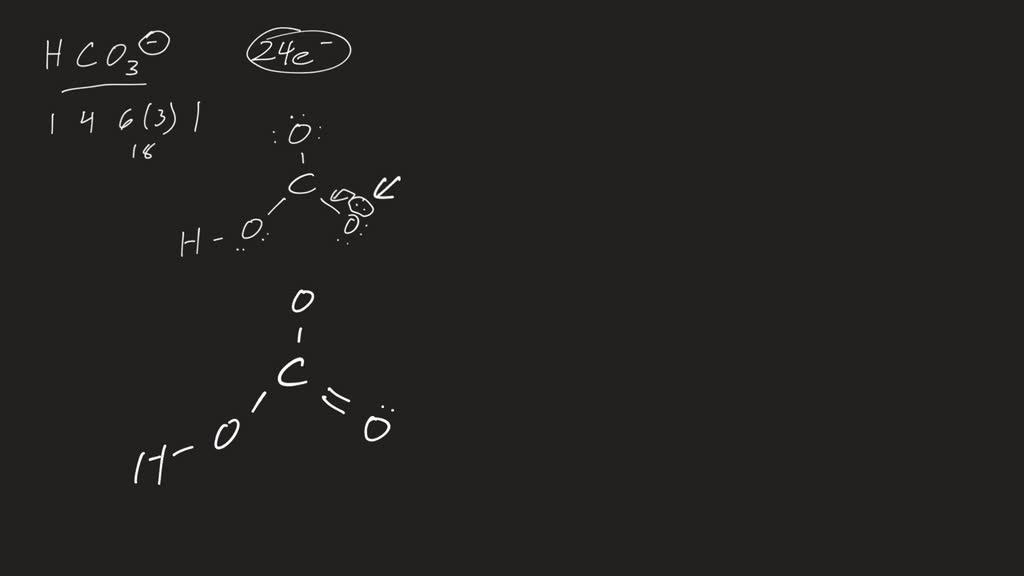
You should put the H CO 3- Lewis structure in brackets with as 1- on the outside to show that it is an ion with a negative one charge. There are a total of 24 valence electrons in H CO 3-. HCO3- Lewis Structure: How to Draw the Lewis Structure for HCO3- Watch on See the Big List of Lewis Structures

Bicarbonate anion hco3 structural chemical Vector Image
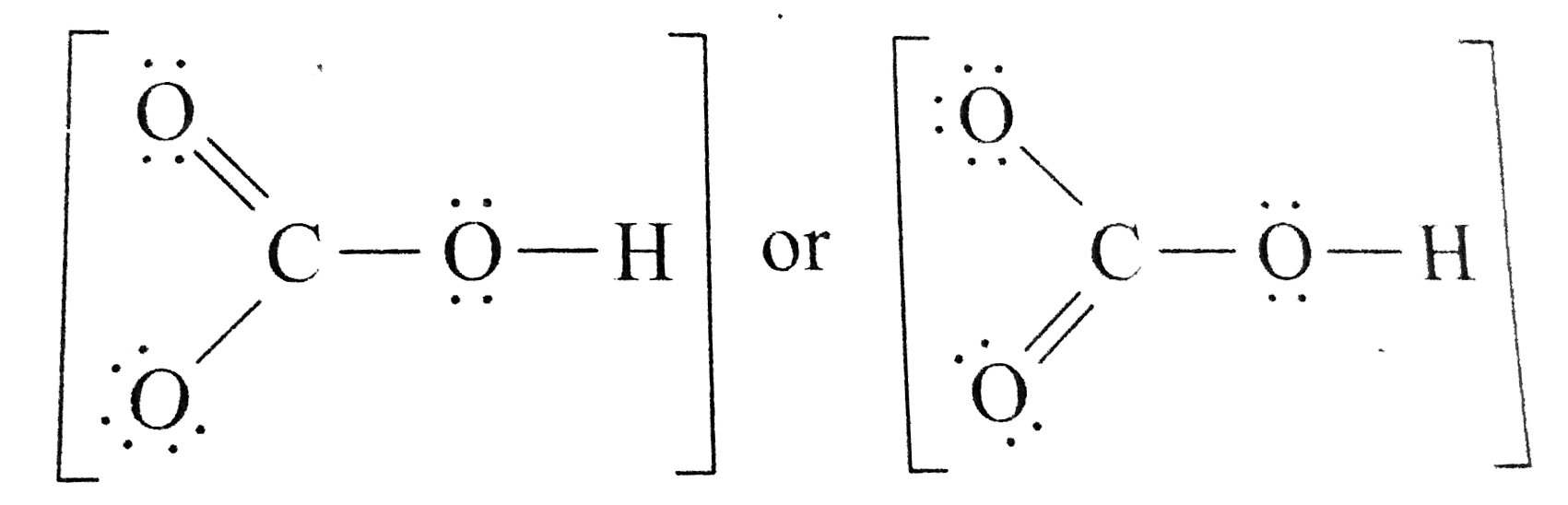
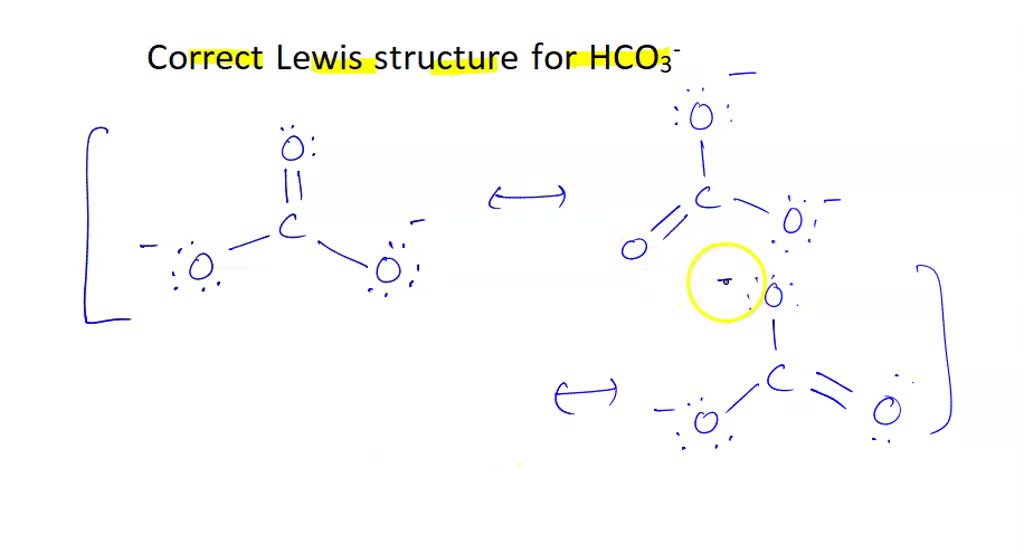
There are two resonance structures HCO3 - (Bicarbonate ion). We start with a valid Lewis structure and then follow these general rules. For the HCO3 - reson.

Hco3lewis Structure
The Lewis structure of HCO3- contains one double bond and three single bonds, with carbon in the center, and hydrogen and three oxygens on either side. The left oxygen atom has three lone pairs, the top oxygen atom and right oxygen atom has two lone pairs, and carbon atom and hydrogen atom do not have any lone pair.
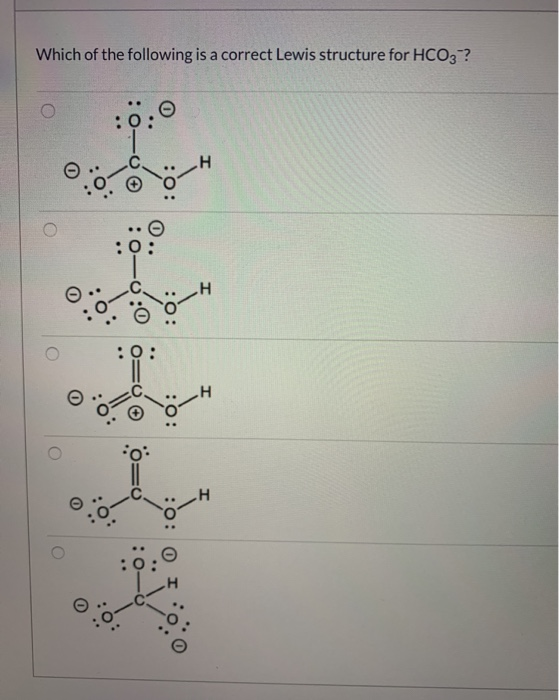
SOLVED Which of the following is the correct Lewis structure for HCO3
How to draw lewis structure of HCO3-? The bicarbonate (HCO3-) ion comprises a carbon (C) atom at the center. It is double-covalently bonded to an oxygen (O) atom at one side and to another O-atom and an OH functional group vis single covalent bonds at the other two sides. There is no lone pair of electrons on the central C-atom.
Lewis Structure For Hco3
Lewis Structures. Page ID. A Lewis Structure is a very simplified representation of the valence shell electrons in a molecule. It is used to show how the electrons are arranged around individual atoms in a molecule. Electrons are shown as "dots" or for bonding electrons as a line between the two atoms. The goal is to obtain the "best" electron.